Approximate Read Time: 14 minutes
“Stem cells don’t rebuild the body. they remind it how to heal. Regeneration begins when biology and movement start working together.”
What You Will Learn
- Stem cells signal and coordinate repair; they don’t directly rebuild damaged tissue.
- Umbilical-cord-derived MSCs show superior potency, safety, and long-term cartilage regeneration outcomes.
- True recovery requires integration between biology initiates healing, but movement completes regeneration.
What Stem Cells Actually Do
When you strip away the hype, stem cells are not a miracle drug and more like an architectural team leader. They don’t build the house. They direct the builders, ensure the blueprints are followed, and keep the work on schedule.
That misunderstanding is that stem cells create new tissue. This is one of the biggest myths in regenerative medicine. It’s a myth that distorts expectations and, perhaps worse, distracts from what actually makes them powerful.
I’ve spent years in professional sport watching athletes chase treatments that promise “rebuilding.” But biology doesn’t work that way. Stem cells don’t construct; they coordinate.
The Signal, Not the Structure
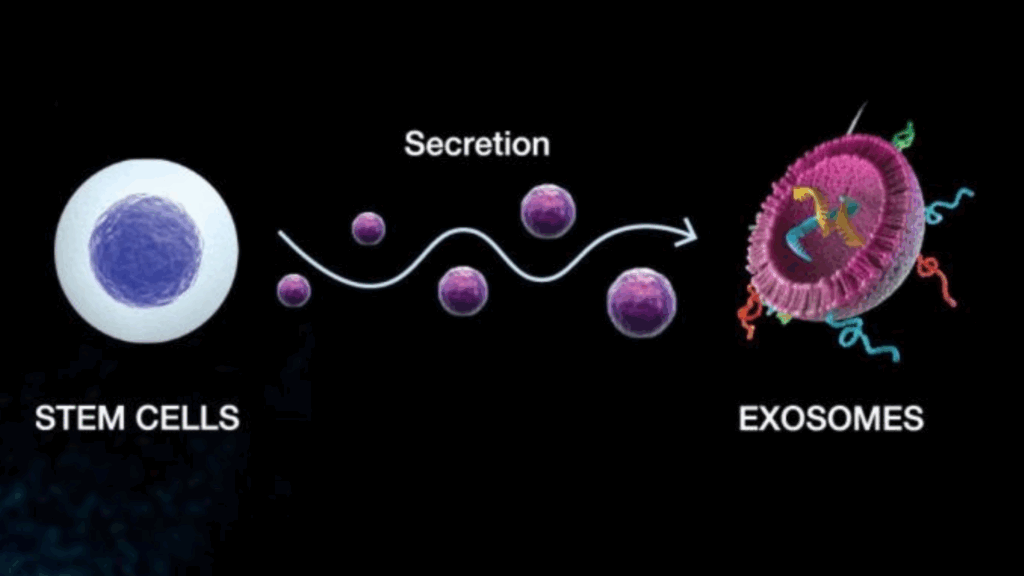
In the language of regenerative medicine, stem cells are paracrine communicators. When introduced into the body, they release tiny packets called exosomes — microscopic vesicles filled with proteins, RNAs, and growth factors. These exosomes act like encrypted messages between cells, telling your body’s own systems how to behave:
- Reduce inflammation.
- Recruit local repair cells.
- Remodel the extracellular matrix.
This process, known as paracrine signaling, explains why stem cells work long after injection. They trigger a cascade, not a replacement. As Gnecchi et al. (2016) described, their power lies in orchestrating a regenerative environment.
Think of it like a symphony. The stem cells are the conductors, not the instruments. They organize the chaos of inflammation into something resembling harmony.
Repair vs. Regeneration: The Subtle Difference That Changes Everything
Repair is what happens when a wound closes. Regeneration is what happens when tissue restores its function. Stem cells help move the body from the first to the second not by becoming new cartilage or tendon, but by restoring order to an environment that’s lost its balance.
In chronic tendon injuries, for instance, we see a familiar pattern: disorganized collagen fibers, erratic blood flow, and weakened tissue. Injecting stem cells doesn’t “grow” a new tendon; instead, they reset the inflammatory rhythm, signaling your own fibroblasts to realign structure and restore tension capacity.
This is why regenerative therapy isn’t just biology rather it’s systems engineering. We’re not replacing parts; we’re recalibrating processes.
Evidence That Refines Expectation
Clinical evidence backs this paradigm. Park et al. (2017) conducted a landmark seven-year trial in which umbilical-cord-derived mesenchymal stem cells (hUCB-MSCs) were implanted into osteoarthritic knees. The results were positive not because cartilage magically grew overnight, but because communication improved.
Arthroscopy revealed that these cells prompted hyaline-like cartilage formation, a type more structurally durable than the fibrocartilage produced by microfracture procedures. MRI confirmed that the cartilage retained its integrity for years, with no tumorigenesis or abnormal bone growth (Park et al., 2017).
A 2022 meta-analysis by Lee et al. pooled results from over 500 patients and found that umbilical-cord-derived stem cells led to consistent, statistically significant improvements in pain (VAS), function (WOMAC, IKDC), and cartilage quality outperforming bone marrow aspirate and adipose-derived cells in several subgroups (Lee et al., 2022)
Across studies, the pattern is the same: stem cells enhance the body’s communication network. They calm overactive inflammation, recruit the right players, and guide the reconstruction process.
The Takeaway So Far
The next time someone claims stem cells “grow new cartilage” or “rebuild a knee,” it’s worth remembering that what they really do is restore the blueprint. Their work happens in the language of signaling molecules and immune modulation, but powerful enough to reshape how tissue behaves.
In that way, they’re less like replacement parts and more like software updates: rewriting the code that tells your biology how to heal.
Not All Stem Cells Are Created Equal
When most people talk about “stem cells,” they imagine a single, monolithic therapy one cell type with universal power. In reality, stem cells are more like a sports team: they share a title, but their capabilities depend entirely on their background, conditioning, and environment.
The industry loves to blur those lines. Bone marrow aspirate, fat-derived cells, umbilical cord tissue each gets tossed under the same umbrella term. Yet their biology, potency, and clinical outcomes tell very different stories.
The Age Problem
Bone-marrow-derived mesenchymal stem cells (BM-MSCs) were early pioneers of regenerative therapy. They’re autologous meaning taken from a patient’s own hip, concentrated, and re-injected. The logic feels comforting: use your own cells, avoid rejection.
But biology plays a cruel trick. By the time a person needs regenerative therapy, typically in their forties, fifties, or beyond, those same stem cells have already aged. Research consistently shows that the number and quality of stem cells in bone marrow decline dramatically with age (Veronesi et al., 2013). A 60-year-old’s bone marrow simply doesn’t contain enough viable MSCs to orchestrate meaningful repair.
Adipose-derived stem cells (AD-MSCs) sidestep some of that problem. They’re abundant and easy to harvest. Yet abundance isn’t the same as performance. Adipose cells tend to prioritize fat-related differentiation, and their chondrogenic potential, the ability to direct cartilage-like tissue, is markedly weaker than younger, developmentally “fresher” cells (Zhang et al., 2011).
This is where umbilical-cord-derived MSCs (UC-MSCs) become the gold standard. These cells are ethically sourced from healthy donor cords after birth non-invasively, without harm to mother or child and they behave as if they’ve just entered the world. They replicate quickly, signal strongly, and carry a unique immunological signature that lets them function safely across genetic lines (Roura et al., 2015).
In simple terms: they’re the youthful blueprint your own cells used to be.
FREE DOWNLOAD
Start Optimizing Your Stem Cell Recovery Today with this Recovery Guide!
Youth as Therapeutic Currency
In regenerative medicine, youth isn’t a metaphor rather it’s measurable. UC-MSCs show higher proliferation rates, longer telomeres, and greater secretion of anti-inflammatory cytokines compared with their bone marrow counterparts (Zhang et al., 2011; Lee et al., 2022). They produce larger volumes of exosomes, those biochemical text messages that regulate local inflammation and healing.
This matters clinically. When Park et al. (2017) implanted a composite of human umbilical cord blood–derived MSCs and hyaluronic-acid hydrogel into patients with advanced osteoarthritis, the regenerated tissue looked and behaved like true hyaline cartilage not scar-like fibrocartilage. Even more striking, the improvements in pain and function persisted for seven years, with no tumor formation or immune reaction.
That kind of durability is rare in orthobiologic research. Most interventions fade as inflammation returns; UC-MSCs appear to change the conversation at the molecular level, not just the symptom level.
A later meta-analysis pooling data from over 500 patients confirmed that pattern. Across trials, UC-MSCs delivered significant improvements in pain (VAS ↓ ≈ 4.8 points), function (IKDC ↑ ≈ 33 points; WOMAC ↓ ≈ 31 points), and MRI-verified cartilage quality all with a strong safety record and no severe adverse events (Lee et al., 2022).
The message was clear: the younger the cell, the stronger the signal.
Ethics, Safety, and Regulation
For many clinicians, “donor-derived” still raises an eyebrow. The assumption is a risk of rejection, infection, or uncontrolled growth. Yet the data say otherwise.
UC-MSCs are hypoimmunogenic, meaning they express minimal surface markers that trigger immune attack (Aggarwal & Pittenger, 2005). In both pre-clinical and human trials, they’ve shown no cases of graft-versus-host reaction, tumorigenesis, or unwanted ossification (Park et al., 2017; Ha et al., 2015).
Even after seven years of follow-up, participants in Park’s study reported no progression to knee replacement and maintained stable function scores. The transplanted cells gradually disappeared within months and triggered the cascade that instructed the body to repair itself (Ha et al., 2015).
That self-limiting lifespan is precisely what makes them safe: they act, signal, and then exit.
From an ethical standpoint, umbilical-cord sourcing represents a quiet revolution. Unlike embryonic stem cells, which carry complex moral debates, UC-MSCs are collected from tissue that would otherwise be discarded. Every donor is screened, every batch is expanded under Good Manufacturing Practice conditions, and each lot is genetically characterized before clinical use (Park et al., 2017).
It’s regenerative medicine without the ethical baggage.

Comparative Reality: BMAC vs UC-MSC
The simplest way to appreciate the difference is to look at outcomes head-to-head.
A 2021 comparative study by Yang et al. found that, following high tibial osteotomy, patients receiving umbilical-cord MSCs showed more complete and higher-quality cartilage regeneration than those receiving bone marrow aspirate concentrate (BMAC), despite similar pain relief (Yang et al., 2021).
Meta-analyses echo the result: UC-MSCs either match or outperform BMAC across pain and function metrics, while demonstrating superior structural repair under arthroscopy (Lee et al., 2022).
The takeaway is practical, not ideological. Autologous cells are useful for small defects and younger patients; donor-derived UC-MSCs seem to excel when biology has already slowed down. They offer consistency—a uniform cell population, potent secretory profile, and predictable outcomes.
The Global Frontier
Because the U.S. FDA still classifies expanded allogeneic MSCs as biologic drugs requiring Investigational New Drug approval, most of the advanced clinical work has taken place abroad in Panama, Japan, and other research-forward jurisdictions. Centers like Auragens in Panama City have become hubs for this science, integrating high-purity UC-MSC protocols with structured pre- and post-rehabilitation programs.
That integration of cell biology meeting biomechanics is where regenerative medicine grows up. It’s also where I’ve focused much of my recent work: building frameworks that don’t stop at injection, but translate cellular potential into functional capacity.
The Big Picture
Stem-cell therapy isn’t a contest between products; it’s an evolution of philosophy.
Bone-marrow and adipose cells taught us what’s possible. Umbilical-cord cells are teaching us how to make it reproducible.
They represent the next iteration of regenerative care. A standardized, potent, and ethical choice ready to be paired with movement science, strength, and recovery in a single continuum.
Why “Stem Cells Replace Good Rehab” Is the Most Dangerous Myth of All
Every field that touches human performance eventually runs into the same trap: mistaking the spark for the engine. In regenerative medicine, the spark is the stem-cell injection and the moment biology seems to take over. The engine is everything that happens afterward: circulation, loading, nutrition, and movement.
When patients believe stem cells do the work, they often stop doing their work. That misunderstanding costs more than money; it costs adaptation.

Biology Starts the Fire; Mechanics Fan It
Stem cells set the biological stage by modulating inflammation and recruiting cells to promote healing. Yet those signals are profoundly influenced by mechanical environment. Motion, load, and oxygen regulate how the newly activated cells behave (Butler & Moseley, 2003).
In animal models, MSCs exposed to controlled mechanical stress show enhanced differentiation into ligament and cartilage-forming cells, whereas immobilized tissue often produces fibrocartilage—strong enough to scar, too weak to perform (Ha et al., 2015; Lee et al., 2022).
Movement, in other words, is molecular. After a regenerative procedure, the body isn’t fragile—it’s plastic. It’s listening for new instructions.
The 12-Week Window: Where Regeneration Meets Rehabilitation
The LEGACY program is designed around this biological truth. The framework unfolds in three phases that parallel stem-cell physiology:
1. Preparation Phase (Weeks −4 to 0)
Before the procedure, the goal is preparation—improving mitochondrial function, circulation, and tissue oxygenation through aerobic conditioning and mobility work. This lowers systemic inflammation and enhances stem-cell homing once they’re delivered.
2. Integration Phase (Weeks 0 to 6)
Immediately after treatment, biology leads. Low-load movement, gentle compression, and recovery modalities preserve muscle signaling without over-stressing the tissue undergoing regeneration. Stem-cell activity peaks during this window, and mechanical input directs their differentiation into the right lineage (Park et al., 2017).
3. Rebuild Phase (Weeks 6 to 12)
Here, exercise reclaim the stage. Progressive resistance, proprioceptive retraining, and controlled plyometrics teach the regenerating tissue to tolerate load. Circulation and neuromuscular drive now determine how well those new cells integrate.
Patients who treat this window as sacred—combining biology with movement—see longer-lasting outcomes. Those who don’t often see biology’s gift fade back to baseline.
Learn more About the LEGACY Membership. The World’s Most Complete Regenerative Performance System.
Regeneration as a Systems Model
The literature supports this integrated approach. In the seven-year follow-up of umbilical-cord-derived MSC implantation, participants maintained improved knee scores only when paired with structured rehabilitation emphasizing quadriceps activation and gradual load exposure (Park et al., 2017). Similar trends appear in post-ACL and tendinopathy data: tissue quality improves only when new collagen is trained into alignment.
Stem cells don’t “replace” rehab; they upgrade the body’s responsiveness to it. They provide fresh cellular clay, but movement is the sculptor.
Metrics That Matter
Regenerative progress should be measured not just by MRI but by capacity metrics:
- VO₂ max for systemic efficiency.
- DEXA or ALMI for lean mass integrity.
- Strength profiling and force-plate data for neuromuscular symmetry.
These metrics borrowed from performance science quantify whether the new tissue behaves like an athlete’s tissue or merely looks like it on imaging. Biology provides the substrate; performance testing validates the integration.
Beyond the Needle
The great irony of regenerative medicine is that its success depends on the same fundamentals that built sport: sleep, nutrition, strength, and stress management. Stem cells amplify whatever environment they’re placed in healthy or chaotic.
Inject them into systemic inflammation, poor sleep, and inactivity, and they’ll mirror that dysfunction. Inject them into a body primed through aerobic base, clean nutrition, and structured load, and they’ll behave more optimally.
Regeneration is not about outsourcing healing. It’s about creating the conditions where healing becomes inevitable.
Closing Reflection: The Starting Line, Not the Finish
Stem-cell therapy doesn’t end with a syringe; it begins there. It is the first lap of a race that includes biomechanics, recovery, and psychology.
Biology sets the foundation. Movement writes the blueprint. Time, discipline, and intelligent stress turn both into architecture.
The promise of regenerative medicine isn’t that it replaces effort, it’s that it rewards it.
Stem cells help you get back to the starting line. Rehabilitation and performance training decide how far you’ll go next.
Conclusion
- Stem cells signal; they don’t build.
- Umbilical-cord MSCs are youthful, potent, and safe, proven to sustain results for up to seven years (Park et al., 2017; Lee et al., 2022).
- Regeneration succeeds only when biology and biomechanics operate in concert.
- The LEGACY framework turns that concert into a system—where medicine meets movement.
Read More Like This
Related Podcasts
References
Lee, D. H., Kim, S. A., Song, J.-S., Shetty, A. A., Kim, B.-H., & Kim, S. J. (2022). Cartilage regeneration using human umbilical cord blood-derived mesenchymal stem cells: A systematic review and meta-analysis. Medicina, 58(12), 1801. https://doi.org/10.3390/medicina58121801
Park, Y.-B., Ha, C.-W., Lee, C.-H., Yoon, Y.-C., & Park, Y.-G. (2017). Cartilage regeneration in osteoarthritic patients by a composite of allogeneic umbilical cord blood-derived mesenchymal stem cells and hyaluronate hydrogel: Results from a clinical trial for safety and proof-of-concept with seven years of extended follow-up. Stem Cells Translational Medicine, 6(2), 613–621. https://doi.org/10.5966/sctm.2016-0157
Yang, H.-Y., Song, E.-K., Kang, S.-J., Kwak, W.-K., Kang, J.-K., & Seon, J.-K. (2022). Allogenic umbilical cord blood-derived mesenchymal stromal cell implantation was superior to bone marrow aspirate concentrate augmentation for cartilage regeneration despite similar clinical outcomes. Knee Surgery, Sports Traumatology, Arthroscopy, 30(1), 208–218. https://doi.org/10.1007/s00167-021-06649-2





