Approximate Read Time: 18 minutes
Osteoarthritis isn’t a cartilage problem. The latest research reveals the problem is more than just a joint – it’s the the whole body.
What You Will Learn
- The “wear & tear” model is incomplete and outdated.
- Osteoarthritis is compounded with metabolic insufficiency and hormones.
- Improve treatment interventions with GLP-1s or stem cell therapy.
An Outdated Model
Most people think osteoarthritis works like a worn-out tire. Years of use gradually grinding down the cartilage until eventually there’s bone rubbing on bone. This mental model shapes everything about how we treat it: we take X-rays looking for cartilage loss, inject the joint with steroids or lubricants, manage pain with medications, and eventually replace the joint when it gets bad enough.
A 2025 research review published in Nature Reviews Rheumatology challenges this entire framework. The paper presents evidence that osteoarthritis isn’t a local cartilage problem at all. It’s a whole-body disease involving broken metabolism, chronic inflammation, fat tissue sending bad signals, and organs that have stopped communicating properly. The joints are just where the symptoms show up.
This distinction matters. Understanding OA as a systemic problem explains why decades of cartilage-focused drug development has failed, why your pain level doesn’t match what shows up on imaging, and why some people develop severe arthritis after an injury while others with the exact same injury don’t. More importantly, it reveals better ways to treat it that focus on strategies that address your whole system, not just your joint.
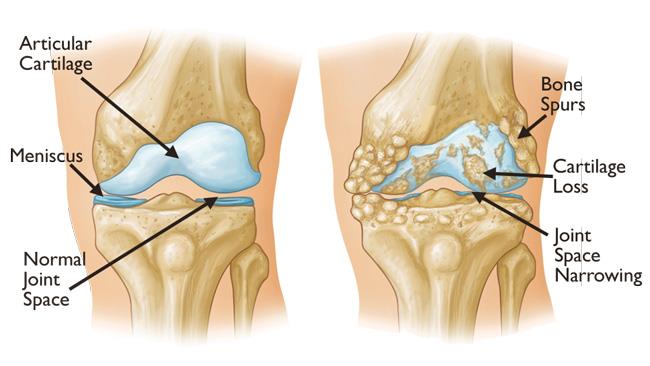
Why the Cartilage Story Never Made Sense
The traditional view of OA came from studying people with end-stage disease: destroyed cartilage, bone spurs, narrowed joint space. Since cartilage can’t repair itself and it serves as the cushion between bones, researchers assumed it was ground zero for the disease. That led to decades of research trying to stop cartilage breakdown.
Here’s what happened: drugs have not resolved the issue. There are medications that help slow down some of these processes or anti-inflammatory drugs to help manage some of the symptoms.
The 2025 review identifies the core problem: these trials treated OA as one disease when it’s actually an umbrella term for many different problems that all end up damaging joints. Injury, genetics, sex hormones, obesity, aging, joint misalignment, and metabolic dysfunction each creates vulnerability through different biological mechanisms. A drug targeting one pathway can’t fix a disease driven by five others.
Imagine prescribing the exact same ACL rehab protocol to every patient regardless of how they got injured, what surgery they had, their psychological state, their training background, or when they need to return to sport. You’d fail most of them because you’re ignoring everything that makes them different. That’s essentially what arthritis drug trials did. They enrolled everybody with joint pain and cartilage damage, regardless of what was actually driving their disease.
What the Research Actually Shows: The Whole Body Talks to Your Joints
The Nature Reviews paper compiles evidence showing that your joints don’t exist in isolation. Rather, they’re constantly receiving signals from the rest of your body. When your metabolism breaks down, when fat tissue starts sending inflammatory hormones, when your gut bacteria shift, when your stress hormones stay elevated – your joints are listening. When enough bad signals converge, cartilage breaks down not because it’s mechanically worn out, but because the biological environment has become unfavorable.
Fat Tissue Talks to Joints (And Not About BodyWeight)
The strongest evidence comes from studying obesity. People with obesity develop arthritis not just in their knees and hips, which makes sense if it’s about load, but also in their hands. Your hands don’t care how much you weigh. This single observation questions the wear-and-tear hypothesis.
The mechanism involves hormones secreted by fat tissue. Leptin, which normally tells your brain you’re full after eating, is one of the main culprits. The research shows that mice genetically engineered to lack leptin are protected from arthritis even when they’re massively obese. On the flip side, giving leptin to mice without any excess fat is enough to trigger joint degeneration. The hormone itself, not the weight, may be a driver to the disease.
Think of leptin like a radio signal your fat tissue broadcasts to the rest of your body. When that signal gets too loud (as it does with obesity), it doesn’t just reach your brain, it reaches your joints, turning on inflammatory pathways and breaking down cartilage.
Here’s where pain comes in: leptin drives joint pain independently of how much damage exists. Studies of people with hand arthritis show that those with obesity report more severe pain in their knees, hips, and hands. This relationship runs through leptin levels, not body weight. This explains why bariatric surgery reduces not just lower body joint pain but hand pain too, and does so almost immediately after surgery, before people have lost significant weight.
The clinical implication of this is simple: pain isn’t simply proportional to cartilage damage. Metabolic factors create nervous system sensitivity that amplifies pain signals even when structural damage is modest. Change the metabolic environment, and pain can improve dramatically without fixing the cartilage.
Inflammation: Smoldering Embers Before the Fire
For years, researchers debated whether OA even involved inflammation because it didn’t look like rheumatoid arthritis with its hot, swollen joints and aggressive immune attack. Some argued it shouldn’t be called osteoarthritis at all because “arthritis” meant inflammation, and OA was just mechanical breakdown.
Modern imaging settled that debate. MRI and ultrasound reveal widespread inflammation in arthritic joints demonstrated by fluid accumulation, inflamed synovial lining, fat pad irritation, and bone marrow stress reactions. People with persistent inflammation show faster cartilage loss and worse pain.
Critically, this inflammation often appears before obvious arthritis develops and can occur in people with no history of joint injury. The research shows inflammatory changes in people with early metabolic problems. This metabolic influence suggests systemic inflammatory signals prime joints for damage before mechanical factors become relevant.
Consider this analogy: inflammation isn’t the fire you see when the joint is already destroyed, rather it’s the smoldering embers that make everything flammable. The cartilage, bone, and joint lining are sitting there ready to ignite. Then a match gets introduced (an injury, overload, aging process) and the whole thing goes up.
Your Gut Bacteria Are Part of the Conversation
Recent research reveals another whole-body connection: your gut microbiome. Studies show that specific patterns of gut bacteria associate with arthritis, and mice raised without any gut bacteria show less joint damage after injury. Transplanting “good” bacteria from healthy mice into arthritic mice reduced disease progression.
The mechanism likely involves bacterial signals that travel through your bloodstream and hormones like GLP-1 (the target of diabetes and weight-loss drugs like Ozempic). A 2025 Science paper showed that OA treatment through GLP-1 drugs works by targeting gut signaling pathways. This is a direct gut-to-joint communication system that has nothing to do with cartilage mechanics.
This explains the excitement around the STEP 9 trial: semaglutide (Ozempic/Wegovy) showed greater knee pain improvement than placebo, with benefits appearing rapidly before substantial weight loss. The drug isn’t fixing cartilage; it’s turning down the volume on inflammatory signals throughout your body.
Think of your gut like a radio tower broadcasting signals throughout your body. When the bacteria in your gut shift toward unhealthy patterns, those signals reach your joints and turn up inflammation. Fix the broadcasting station, and you can change what your joints are hearing.
Sex Differences Run Deeper Than Hormones
Women have higher rates of arthritis, worse symptoms, and different pain patterns than men. The traditional explanation pointed to menopause and estrogen loss, but the research shows sex differences are more complex.
Sex differences in cartilage biology show up before menopause and persist even when estrogen is removed in animal studies. This means they’re not purely hormonal. Emerging research points to chromosomal differences that drive immune activation in women independently of sex hormones.
Metabolism compounds these differences. Lifetime arthritis risk is highest in women with obesity and lowest in men without obesity. This is a complex interaction between biological sex, fat tissue metabolism, and aging. Belly fat (not overall body fat) associates most strongly with knee pain, and this effect is mediated through inflammatory proteins and leptin.
The clinical takeaway is that sex-specific differences in OA aren’t just about joint shape or hormonal cycles. They reflect fundamental differences in immune function, metabolic regulation, and how the nervous system processes pain. These are differences that must be considered in treatment approaches.
Why Mouse Studies Haven’t Led to Human Treatments
If systemic factors drive arthritis, why have decades of research using mice with obesity, metabolic problems, and joint injuries failed to produce effective drugs?
The Nature Reviews paper identifies critical mismatches between how studies are designed and the disease we’re trying to treat:
- Most studies use young male mice. This is like training all your physical therapists on 25-year-old male athletes and then asking them to treat 55-year-old women with multiple health problems. The biology is completely different. The clinical OA population is mostly middle-aged women with metabolic issues and other chronic conditions. Testing drugs in young males tells us about one narrow slice of biology, not the diverse disease we see in the clinic.
- Studies measure cartilage damage at fixed time points, missing the early disease. Most research looks at cartilage under the microscope 8-12 weeks after injury. But metabolic changes, immune activation, and pain development happen weeks earlier before you can see obvious joint damage. By focusing on end-stage pathology, researchers miss the intervenable mechanisms. It’s like only studying house fires after the building has collapsed instead of when smoke first appears.
- Pain measurement is a recent addition. Until the past few years, animal OA models rarely measured pain behavior. They just looked at cartilage damage. Yet pain is the main reason people seek treatment. Studies now show that structural damage and pain can be separated. For example, female mice show similar pain severity to males despite less cartilage destruction.
- Single-joint focus misses systemic patterns. Most models injure one knee and measure outcomes only in that joint. But systemic OA often hits multiple joints simultaneously. Diet-induced obesity models show arthritis in both knee and shoulder (but not hip) in rats. This suggests joint-specific vulnerability to body-wide signals that would be completely missed studying knees alone.
The consequence of single-joint drugs that work in young male mice with isolated knee injuries don’t necessarily translate to middle-aged women with metabolic syndrome, multi-joint involvement, widespread pain sensitivity, and comorbidities. The models aren’t broken, rather they’re just answering different questions than clinicians need answered.
Treating the System, Not Just the Joint
If arthritis is a whole-body disease, rehabilitation must address whole-body factors and not just deliver local joint interventions. This shifts the question from “how do I protect cartilage?” to “how do I create a biological environment where joints can stay healthy?”
Your Metabolism Is Part of Your Musculoskeletal Health
The research makes clear that metabolic dysfunction such as insulin resistance, unhealthy cholesterol, visceral fat, or chronic inflammation directly drives joint breakdown and pain. This means that for many people with arthritis, improving metabolic health is a primary treatment, not a nice-to-have bonus.
The practicality of this systemic perspective is that we should check metabolic health in all patients with OA, especially those under 65. A 2025 study showed that fat tissue starts showing aging signatures in people’s 40s, which is well before most show obvious metabolic problems. Early metabolic disturbance creates joint vulnerability that manifests years later as arthritis.
Body composition matters more than weight. People with sarcopenic obesity, low muscle mass combined with high body fat a.k.a. “skinny fat”, have worse arthritis outcomes and higher disease rates, even when their weight looks normal. A trial called POMELO showed that combining nutrition, resistance training, and self-management improved muscle, mobility, and quality of life better than weight-loss-focused approaches alone.

Think of it like soil quality for a garden. You can try to prop up struggling plants with spray treatments, but if the soil is toxic with poor nutrients, nothing thrives. Fix the soil and everything grows better.
If we tie this back to the 3P Model – Principles, Process, and Plans – then this systemic perspective is fairly simply.
- Principle = metabolic health influences joint health systemically.
- Process = assess body composition and metabolic markers (not just scale weight), screen for diabetes risk and inflammation.
- Plans = integrate resistance training to preserve muscle, nutrition strategies targeting inflammation and insulin sensitivity, cardiovascular work for systemic metabolic improvement.
Want to learn systems-based approaches to injury rehabilitation and performance?
Load Management Depends on System Vulnerability
Understanding OA as systemic disease doesn’t eliminate the importance of mechanical load, rather it contextualizes it. Joints become vulnerable when whole-body factors create hostile environments. Mechanical load then acts as accelerant rather than root cause.
This explains what the research calls the “multi-hit hypothesis”: why some people develop severe arthritis after ACL injury while others with identical injuries don’t. The joint injury is one hit; systemic factors (obesity, metabolic syndrome, genetics) are additional hits that determine whether pathology progresses.
Think of it like a bridge. A structurally sound bridge with good materials can handle heavy traffic. But if the steel is corroded (metabolic dysfunction), the cables are frayed (chronic inflammation), and the foundation is cracked (genetic vulnerability), even normal traffic causes problems. Adding more load doesn’t help, but the real issue isn’t the traffic, but it’s the bridge’s underlying integrity.
Load management therefore needs individualization based on systemic vulnerability. An athlete with excellent metabolic health, low inflammation, and favorable genetics can likely tolerate more aggressive loading progressions post-injury than someone with elevated inflammatory markers. This is true even if both have identical joint damage and injury severity.
When progressing loading in rehab, consider not just local joint capacity (strength, range of motion, tissue healing) but systemic factors that modify tissue resilience. An executive with poor sleep, high stress, inflammatory diet, and elevated blood sugar needs different loading progressions than an athlete with optimal recovery and metabolic health despite similar joint pathology.
Look at All the Joints, Not Just the Painful One
If systemic factors drive arthritis vulnerability, assessing only the symptomatic joint misses the bigger picture. The research advocates for checking multiple joints rather than isolated single-joint evaluation.
When evaluating knee pain in someone with obesity and metabolic problems, check their hands, hips, shoulders, spine and not just the painful knee. Multi-joint involvement suggests systemic drivers rather than isolated mechanical overload. This completely changes treatment strategy from local joint protection to whole-system optimization.

Movement quality matters through a different lens too. Rather than thinking “poor biomechanics wear down cartilage,” consider that inefficient movement patterns increase energy cost and metabolic demand. Improving movement efficiency reduces systemic stress, not just local joint forces.
Pain and Structure Can Be Addressed Separately
One of the most clinically useful insights from the research is that pain and structural damage in arthritis are partially separable, driven by overlapping but distinct mechanisms. This means treatments can reduce pain without changing joint structure, and fixing structure may not resolve pain if systemic pain drivers remain unaddressed.
The research identifies pain-susceptible profiles characterized primarily by nervous system sensitization, psychological factors, comorbidities, and systemic inflammation. In the MOST cohort, people with high psychological burden, multiple health conditions, and pain sensitivity, but low structural damage reported the worst pain levels.
Think of pain like a smoke alarm. The alarm going off (your pain) doesn’t necessarily correlate with the size of the fire (cartilage damage). Sometimes a small fire (modest joint damage) triggers a hypersensitive alarm (amplified pain). Sometimes a bigger fire doesn’t trigger much alarm response. Fixing the fire won’t help if the alarm itself is malfunctioning.
For patients with severe pain but modest imaging findings, targeting cartilage won’t help. Instead, address sleep, stress, systemic inflammation, metabolic health, psychological factors, and nervous system sensitivity. These aren’t peripheral considerations rather they’re primary drivers.
Interestingly, 20-30% of people report persistent pain after total knee replacement likely because joint damage wasn’t the main pain driver. Systemic factors persist after surgery, continuing to generate pain independent of joint structure.
Where Regenerative Medicine Fits In
Understanding OA as systemic disease creates new context for regenerative approaches like stem cell therapy. Rather than viewing these as attempts to “regrow cartilage,” which isn’t really how they work, the 3P systems framework positions them as biological signal modifiers that can change the joint’s inflammatory environment.
Mesenchymal stem cells (MSCs) work primarily by secreting chemical signals that calm down immune response, reduce inflammation, and promote tissue balance. This mechanism aligns perfectly with the systemic model: rather than structurally rebuilding damaged cartilage piece by piece, MSCs help normalize the hostile biological environment that perpetuates joint breakdown.
FREE DOWNLOAD
Interested in Stem Cell Recovery?
Think of MSCs like a thermostat reset for your joint’s inflammatory environment. When that environment is running too hot (chronic inflammation, immune activation, hostile chemical signals), introducing MSCs doesn’t add new building materials. MSCs turn down the temperature so your own repair systems can work properly.
Stem cell integration makes most sense when timed to modify the biological environment during vulnerable windows such as early post-injury when inflammatory pathways are most active, or in established OA with high systemic inflammatory burden where local treatments alone have failed.
The LEGACY approach reflects these systems understanding:
- Prepare the system (optimize metabolic health, reduce inflammation)
- Restore tissue capacity (strategic MSC administration)
- Perform (progressive loading with continued metabolic optimization).
It’s not about magic cartilage regeneration. It’s about changing the signals that determine whether joints degenerate or maintain health.

Evidence from drug trials provides indirect support. Large cardiovascular studies showed that systemic anti-inflammatory medications, not joint injections, but pills affecting the whole body reduced joint replacement rates by 30-40% when given for 2-4 years. These drugs weren’t fixing cartilage; they were modifying inflammatory signaling throughout the body.
Interventions that modify systemic inflammatory, metabolic, or immune pathways, whether pharmaceutical (GLP-1 drugs, metformin, anti-inflammatories) or biological (stem cells, exosomes), may change arthritis trajectory by addressing root causes rather than downstream cartilage damage.
Let’s Change the Narrative
The conversation shifts from “your cartilage is worn out and there’s nothing we can do” to “your joint became vulnerable due to whole-body factors we can modify.” This reframes OA as manageable chronic condition rather than inevitable decline.
Obesity and excess body weight must be part of the conversation. Fat tissue sends hormone signals to joints and creates an inflammatory environment that accelerates breakdown. By improving systemic health including sleep, nutrition, body composition, and stress then we can directly influence joint health.
Rather than passively accepting decline, they understand concrete actions that modify disease trajectory. The research shows physical function is a better predictor of longevity than almost any lab test, and interventions that improve function extend healthspan.
The Bottom Line
Osteoarthritis isn’t inevitable joint wear-and-tear. The 2025 Nature Reviews paper compiles compelling evidence that OA is whole-body disease involving broken metabolism, chronic inflammation, hormonal signals from fat tissue, immune dysfunction, and organ communication breakdown that happens to show up in joints.
This reframing explains why obesity drives hand arthritis despite no load increase or why pain and imaging findings don’t match. Single-pathway drugs failed yet systemic drugs including like GLP-1 drugs improve joint pain before weight loss.
Most importantly, this perspective gives patients agency. Rather than being told “your cartilage is gone, there’s nothing we can do,” they learn that factors they can control – sleep, nutrition, body composition, stress, metabolic health, movement quality – directly influence whether joints degenerate or stay healthy.
After 15 years with NBA, MLS, and USWNT athletes, here’s what I’ve learned: high performance is about optimizing the biological systems that let tissues adapt to stress. The same principle applies to arthritis. You can’t protect joints from all load. But you can optimize the metabolic, inflammatory, and systemic environment to make those joints resilient.
Watch More Like This
Related Podcasts
References
1. Collins KH, Haugen IK, Neogi T, Guilak F. Osteoarthritis as a systemic disease. Nature Reviews Rheumatology. 2025. doi:10.1038/s41584-025-01332-8
2. Bliddal H, et al. Once-weekly semaglutide in persons with obesity and knee osteoarthritis. New England Journal of Medicine. 2024;391:1573-1583.
3. Collins KH, et al. Adipose-derived leptin and complement factor D mediate osteoarthritis severity and pain. Science Advances. 2025;11:eadt5915.
4. Collins KH, et al. Adipose tissue is a critical regulator of osteoarthritis. Proceedings of the National Academy of Sciences. 2021;118:e2021096118.
5. Yang Y, et al. Osteoarthritis treatment via the GLP-1-mediated gut-joint axis targets intestinal FXR signaling. Science. 2025;388:eadt0548.
6. Godziuk K, et al. Improving muscle function through a multimodal behavioural intervention for knee osteoarthritis and obesity: the POMELO trial. Journal of Cachexia, Sarcopenia and Muscle. 2025;16:e70025.
7. Schieker M, et al. Effects of interleukin-1β inhibition on incident hip and knee replacement. Annals of Internal Medicine. 2020;173:509-515.
8. Heijman MWJ, et al. Association of low-dose colchicine with incidence of knee and hip replacements. Annals of Internal Medicine. 2023;176:737-742.
9. Pan F, et al. Metformin for knee osteoarthritis in patients with overweight or obesity: a randomized clinical trial. JAMA. 2025;333:1804-1812.
10. Carlesso LC, et al. Pain susceptibility phenotypes in those free of knee pain with or at risk of knee osteoarthritis. Arthritis & Rheumatology. 2019;71:542-549.
11. Gloersen M, et al. Associations of body mass index with pain and the mediating role of inflammatory biomarkers in people with hand osteoarthritis. Arthritis & Rheumatology. 2022;74:810-817.
12. Dou DR, et al. Xist ribonucleoproteins promote female sex-biased autoimmunity. Cell. 2024;187:733-749.
13. Hannani MT, et al. Longitudinal stability of molecular endotypes of knee osteoarthritis patients. Osteoarthritis and Cartilage. 2025;33:166-175.
14. Loeser RF, Goldring SR, Scanzello CR, Goldring MB. Osteoarthritis: a disease of the joint as an organ. Arthritis & Rheumatism. 2012;64:1697-1707.
15. Ding Y, et al. Comprehensive human proteome profiles across a 50-year lifespan reveal aging trajectories and signatures. Cell. 2025. doi:10.1016/j.cell.2025.06.047




